No evidence that Y-chromosome differentiation affects male fitness in a Swiss population of common frogs
Male frogs in the same population with Y chromosomes of varying differentiation level from the X have no significant difference in morphometrics, amplexus success and fathering success.
The standard theory of sex chromosome evolution predicts that they will accumulate mutations that are beneficial for one sex and deleterious to the other. Such genes are termed sexually antagonistic.
The reason sex chromosomes attract sexually antagonistic genes is that they spend the majority of their time in one sex. For example, the Y is only found in males, and the X spends twice the time in females (XX) compared to males (XY) in mammals. By localising on sex chromosomes, sexually antagonistic genes can therefore maximise the positive effects to the sex in which they occur most often and minimise the negative effects the same alleles have in the opposite sex.
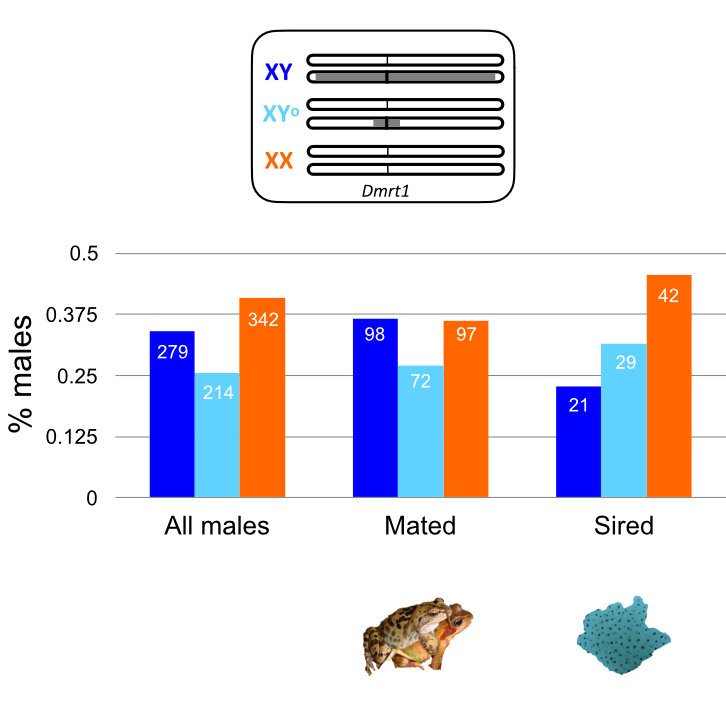
The common frog presents a rare system to test this idea, because populations can be found in which Y chromosomes of varying differentiation to the X exist. We worked on a population with 3 major Y-haplotypes: one which is fully differentiated across its length, one which is only differentiated close to a candidate sex-determining gene, and one which is indistinguishable from the X (XX males). These haplotypes represent different portions of the genome that spend their time in males; the absence of differentiation indicates regular recombination. According to the standard sex chromosome evolution theory, one might expect the fully differentiated haplotype to have accumulated more male beneficial alleles than the other haplotypes found in males.
To do this, we measured frogs in their natural environment during breeding season. It is a short 2-3 week window in which they all travel and meet in a pond at high latitude. They start breeding as soon as some of the pond is not frozen so they can enter it. My first encounter with the field site was in negative temperatures and wind. I was lucky I was not the one to enter the pond in these conditions to catch the frogs.

We looked for biological differences between the Y haplotypes by measuring male fitness. We did this directly, by comparing the proportion of offspring fathered by males with the fully differentiated Y compared to the proportion of free-swimming males and, similarly, the proportion of males with a fully differentiated Y that were found in amplexus with females, compared to free-swimming males. We also measured male size to check whether different Y haplotypes are associated with body size differences.
One can never prove a negative result, and it is possible that larger sampling or a different measurement would identify a difference between the haplotypes. For example, maybe the system is unstable, and variation is only possible from gene flow which introduces lost variation from nearby populations. Or perhaps there are performance differences between the haplotypes in different environments, such as different temperatures, or in their development time, which our study was not particularly good at capturing as it would require sampling over many years.
However, there is a good reason for the standard sex chromosome evolution model not to apply to frogs, which is that sex-chromosome recombination is different than what the model assumes. Frog recombination depends on phenotypic sex and is extremely limited in males to all chromosome ends, including the sex chromosomes. Consequently, a male-determining mutation will immediately restrict recombination to almost the whole chromosome carrying the male-determining allele. This would allow it to capture a male-beneficial mutation and is likely the reason for the spread of a new male-determining mutation. However, it would make it difficult for additional male-beneficial alleles to accumulate, because they can mostly arrive by mutation. Rare recombination events are possible through rare sex-reversal cases when XY individuals develop as females and thus have a female-like chromosome-wide recombination pattern which allows recombination on the Y. The rare sex-reversed individuals, which have been observed in nature, represent a fountain of youth for the Y chromosome, and are likely the source of the semi-differentiated Y haplotypes. Nevertheless, the commonly experiences lack of recombination throughout most of the Y length means that the deleterious effects of no recombination, which are responsible for sex chromosome degeneration according to the standard model, are immediately present in young sex chromosomes in frogs.
It will be interesting to model the evolutionary dynamics of the different forces affecting sex chromosome evolution in systems with achiasmatic meiosis, or meiosis that is extremely restricted to chromosome ends, as in frogs. This type of meiotic recombination is also found in many fishes and Drosophila. It is possible that the sex chromosomes of these species are primarily affected by deleterious mutations over a larger parameter space than they are affected by sexually antagonistic mutations. In this light, it is perhaps not surprising that different frog Y haplotypes do not have obvious differences in fitness.
The published papers is available here.