Reproductive tissue mating response after experimental sexual selection
Experimentally elevated sexual selection increases male manipulation of the female post-mating response, but also makes females poised in readiness for mating and more resistant to male manipulation.
In our new paper on the Drosophila pseudoobscura lines from the Snook lab we investigated effects of experimental sexual selection on mating. This work was based in the Ritchie lab, in collaboration with the Snook lab and Andy Cossins.
We used tissue-specific transcriptomes of reproductive tissues from both sexes. Working with specific tissues is beneficial because it minimises the effects of allometry from the observed response, it can detect gene regulation evolution even if, for example, testes have evolved to be larger under high sexual selection. One disadvantage of using specific tissues from a small insect is that there are fewer cells that provide the RNA, so the dissections required to obtain sufficient RNA took a whole week. We had 4 2-hour blocks of dissections per day.
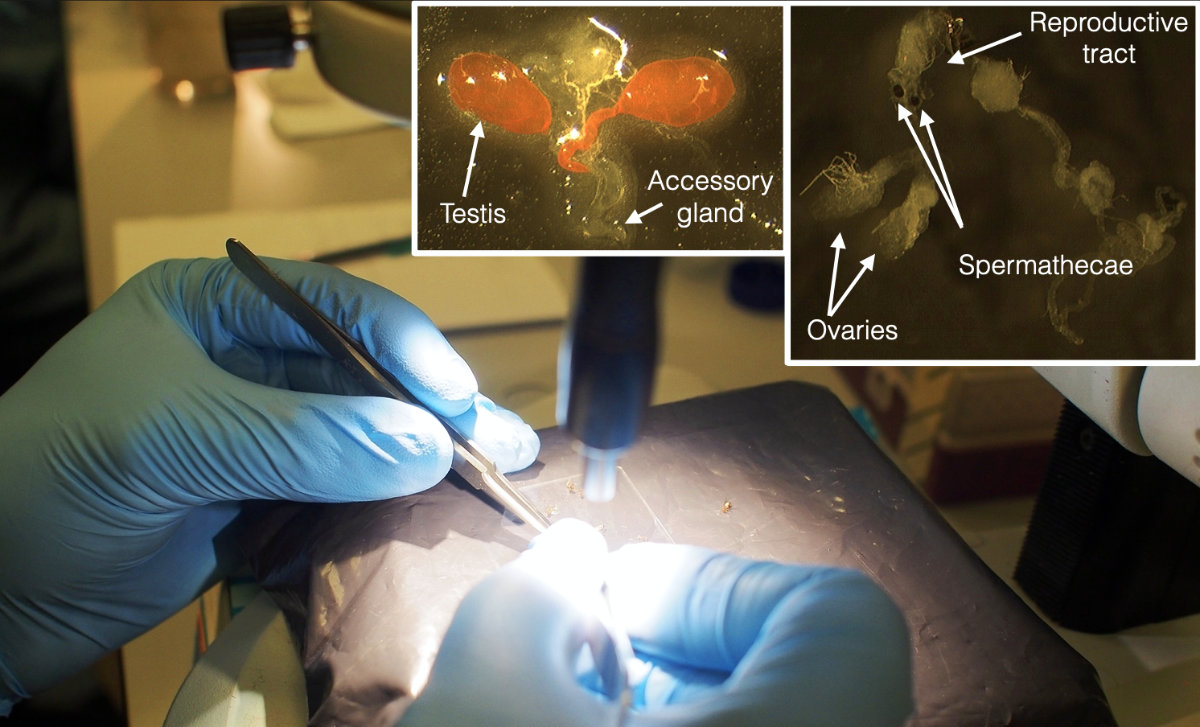
Some dissections involved mated females that had to be prepared first. This made for a very busy week, so that the following week of (lots of) RNA extractions seemed relatively mild in comparison. With so focused physical work events blur and it is difficult to recall the experience. Nevertheless, I remember we were late for the xmas party because we wanted to finish within the year, and that we looked at an inverted microscope after tissue disruption with metal beads, to make sure we had successfully pulverised the very small reproductive tissues, including the hard spermathecae. I also have a vague memory of delivering the samples in ice in person at the Centre for Genomics Research in Liverpool, which prepared and sequenced the libraries.

All this work was before we started analysing the data, which took the most time. Here I will focus on two of our results, both of which involve interactions. But first, a brief note on the experimental design.
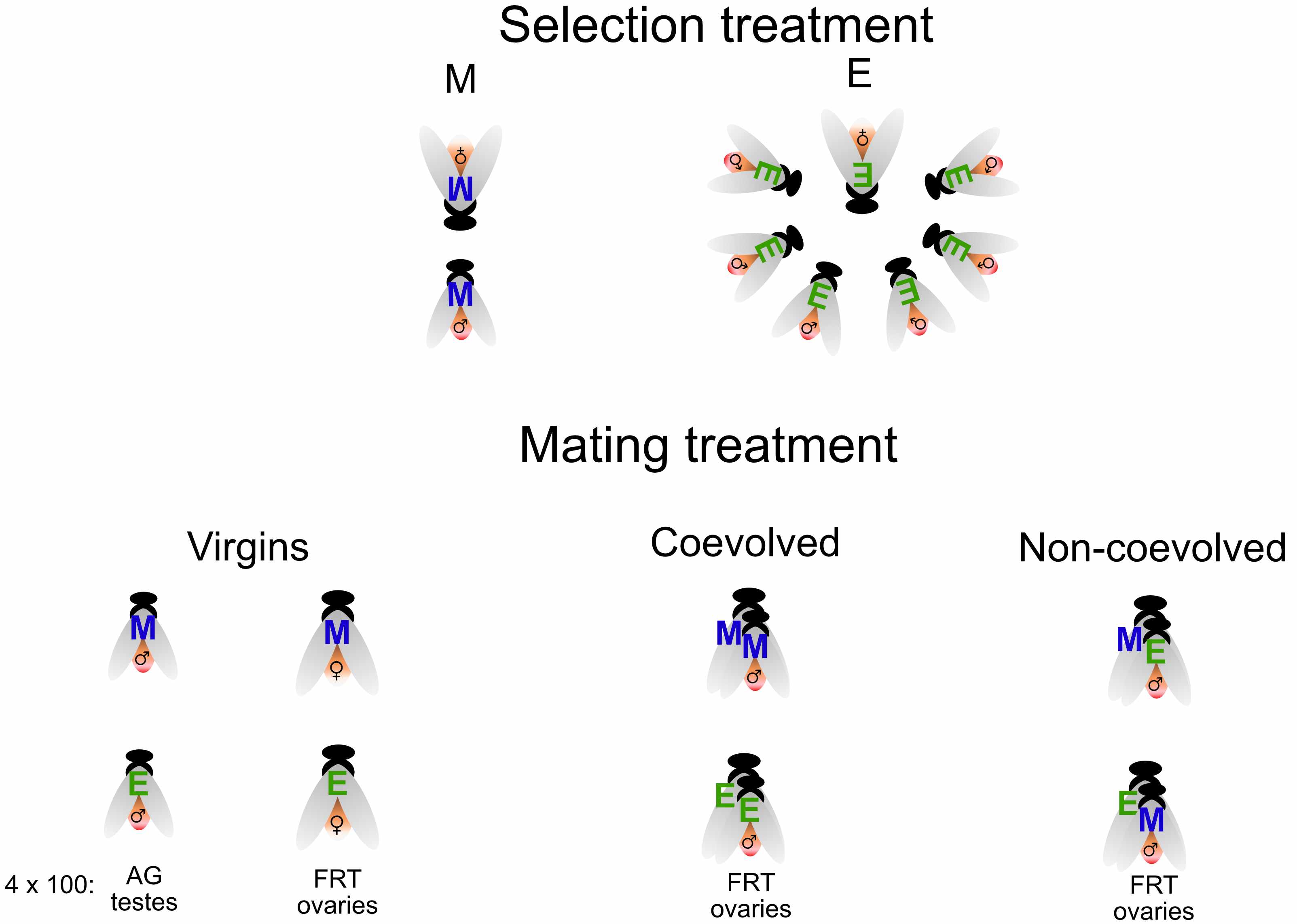
High sexual selection females do not require mating to activate some gene expression
Our experimental design allowed us to look for an interaction between the effect of sexual selection (the overall difference between high and low sexual selection females i.e. E vs M contrast) and the effect of mating (i.e. virgin vs mated contrast). Identifying interactions is quite a standard statistical practice, the main issue is interpreting the result and understanding the biology behind an interaction. One of the most interesting results of the paper is the strong interaction we found between mating status and sexual selection history.
To visualise the interaction, we plotted the difference in expression between virgin and mated females, separately (on the x and y axes) for the E and M females.
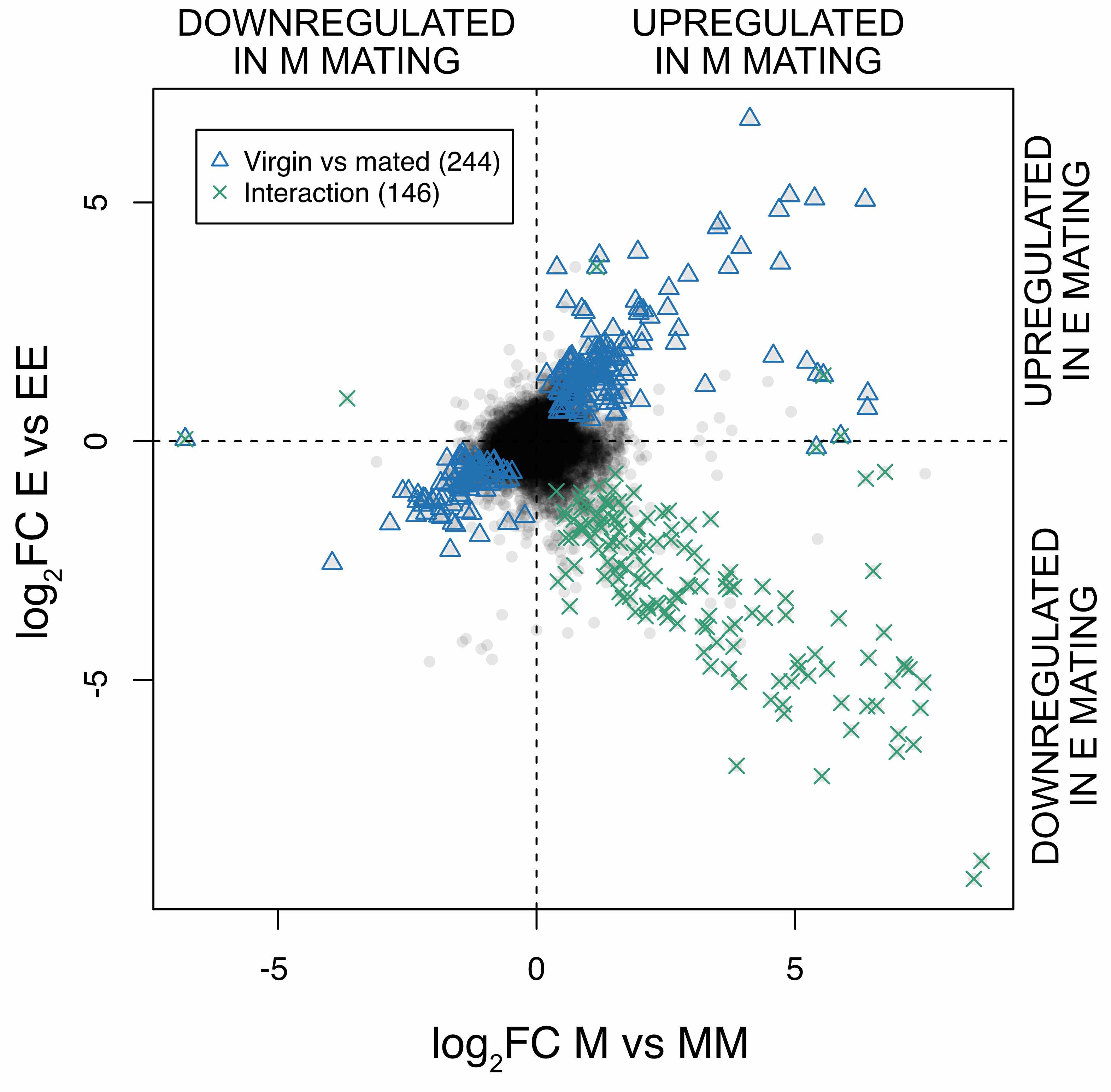
It is quite striking that most of the genes significant for the interaction are in one direction (green points): they are genes expressed in virgin high sexual selection females (E), but only expressed after mating in monogamous females (M).
The result supports previous phenotypic studies suggesting that the high sexual selection females are primed for mating. This might be expected, since they have evolved in an environment where they have the opportunity to mate with different males. The fact that they do not need mating to activate some genes that monogamous females only activate after mating suggests the possibility that there may be a cost to having them expressed as virgins. Their functions are related to the immune system and egg production which makes sense for the female reproductive tract and further support our interpretation that E females are poised for mating.
A similar analysis in ovaries found many genes with a significant main effect of the selection treatment, few genes changing expression based on mating, but no genes significant for an interaction, suggesting the ovariole genes of the two types of female respond the same way to mating. One explanation is that the 6 h after mating in which we sampled for RNA did not change much in terms of gene expression in the ovaries. So perhaps the main conclusion is that it was a very good call to separately analyse the two female reproductive tissues as they show very different results.
High sexual selection males manipulate the female mating response more - but high sexual selection females seem more resistant to this manipulation
We were able to study the effect of different types of male in the female mating response by analysing females of both selection treatments mated with males they had coevolved with and with males they had not. This is a complicated interaction so we plot separately the two types of female.
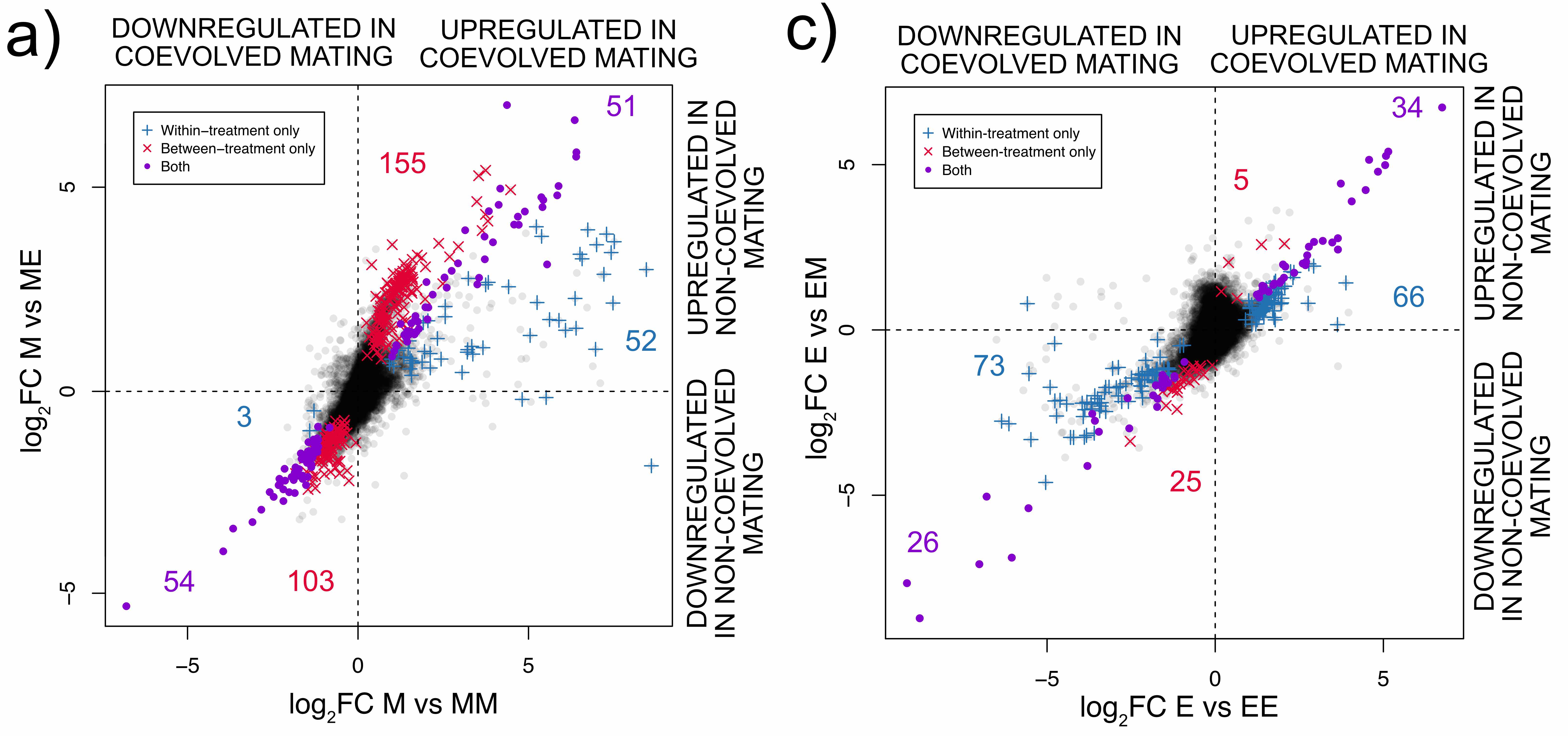
Comparing the number of significant points between the two graphs illustrates that E females do not change the expression of as many genes as M females do, regardless of mating partner. This also supports the suggestion that E females are primed for mating, since they activate genes prior to mating, compared to the M females.
Let us now focus on the two types of male (E or M). Notice that the same type of male has a different colour in the two plots, for example high sexual selection males affect the expression of red genes for a) but blue genes for c). Once you realise this, it is quite clear that E males cause more genes to change expression between virgin and mated M females (a - red genes, 155+103) than E females (c - blue genes 66+73).
The results lead to two intriguing interpretations:
- High sexual selection males manipulate the female postmating response more than low sexual selection males
- Simultaneously, high sexual selection females are more resistant to this manipulation than low sexual selection females.
For the full story, including results on virgin tissues (where male accessory glands and female reproductive tracts had more gene expression changes between E and M treatments than testes and ovaries) and comparisons with proteomic work, see the paper.